Next time you talk on a cell phone, hear a weather report, search the web, or get an MRI, remember the U.S. National Science Foundation helped make that all possible, and more.
Since 1950, NSF has invested in ideas and innovations that have shaped the modern world — pushing the boundaries of what's possible and deepening humanity's understanding of the world.
On this page you can explore NSF-supported research across all fields of science and engineering that power the American economy and touch everyone's lives.
 On this page
On this page
Brought to you by NSF
Explore innovations and discoveries made possible through NSF support.

3D printing
3D printing is spurring a manufacturing revolution in the U.S. thanks to decades of NSF investments.

American Sign Language
NSF funding revolutionized education for deaf and hard-of-hearing individuals in the United States.

Artificial intelligence
NSF funding built the foundation for technologies Americans use every day, such as digital assistants like Alexa and Siri, Face ID, image generators and chatbots like ChatGPT.

Bolstering small business
From artificial intelligence and energy to medical devices and semiconductors, NSF invests in deep-tech startups and small businesses working across all fields of science and technology.

CAPTCHA and reCAPTCHA
The tools that keep badly behaving bots from taking over the internet were invented by NSF-funded researchers.
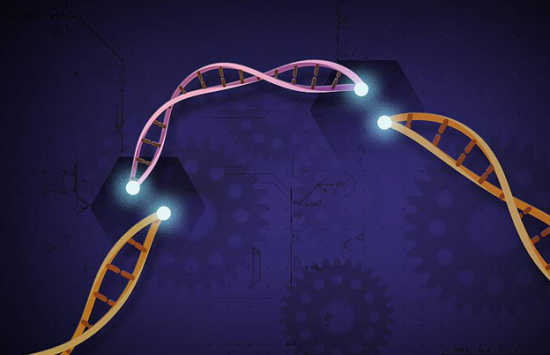
CRISPR
NSF-supported research has advanced one of the sharpest tools in gene technology.
Cybersecurity
For decades, NSF has funded research to protect national and personal security in today's highly connected, digital world.

Deep-ocean exploration
Decades of NSF-funded deep-sea research and subseafloor ocean drilling reveal fundamental knowledge about the entire Earth system.

Detecting gravitational waves
Opened in 1999, NSF LIGO was the first to detect gravitational waves: ripples in space-time.
DNA amplification
Polymerase chain reaction, a lab technique that powers today's biotechnology sector, traces its roots to NSF funding.

Doppler radar
Decades of NSF–funded research have improved the capabilities of the national Doppler radar network.

Duolingo
One of the most popular language-learning tools got its start from NSF funding.

Fusion energy
NSF's decades-long support for fusion R&D is paving the way toward an abundant, reliable power source to meet the world's growing energy demand.
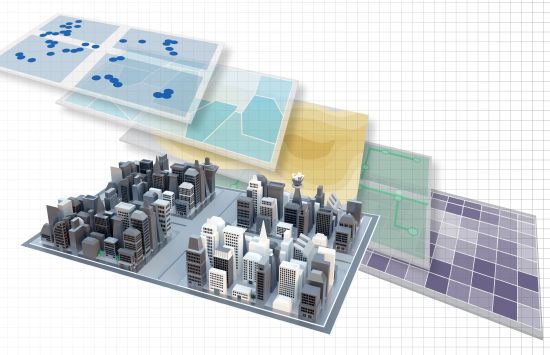
Geographic Information Systems (GIS)
NSF investments helped chart the course for the technologies that use spatial data to make people's lives more convenient.

The internet
Drawing on DARPA's pioneering support for early computer networking projects, NSF funding catalyzed the creation of today's internet.

Kidney matching
Decades of NSF funding for economic and algorithm research has facilitated thousands of lifesaving kidney transplants every year.

LASIK eye surgery
Decades of NSF funding supported the development of bladeless LASIK, which uses a laser instead of a scalpel to reshape a patient's cornea and improve their vision.

Learning beyond the classroom
Throughout its history, NSF has made the excitement of scientific discovery available to everyone by investing in children's TV programming, movies, museum exhibits and apps.
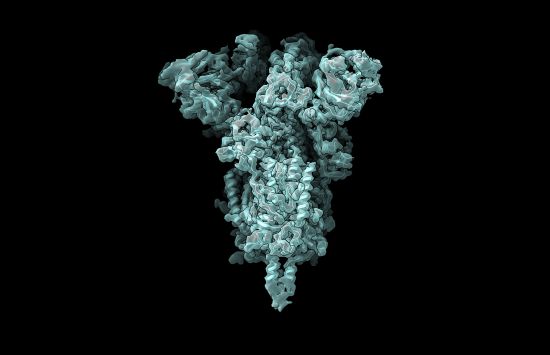
Protein Data Bank: Key to the molecules of life
NSF's decades-long support for the open-access database has facilitated pioneering scientific advancements and medical treatments.
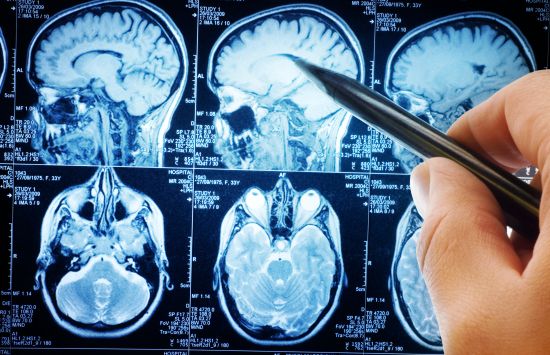
Magnetic resonance imaging (MRI)
One of the most widely used medical imaging technologies is rooted in decades of NSF-powered research.

Natural hazards research: From risk to resilience
NSF-funded research on storms, wildfires and other natural hazards has saved lives, strengthened communities and reduced economic losses.

Seeing black holes
Thanks to NSF support, international researchers have now captured images of two black holes, including the one at the center of the Milky Way galaxy.

Semiconductors
NSF–funded research has catalyzed breakthroughs in semiconductors, the chips that enable virtually all modern technology.
Smartphones
Smartphone technology traces its roots to decades of NSF funding, such as touchscreens, lithium-ion batteries and the internet.

Supercomputers
NSF's investments in cutting-edge supercomputers has catalyzed scientific breakthroughs and sharpened the nation's competitive edge.
NSF investments
Learn about NSF's ongoing investments and impacts in major areas of research and innovation.
NSF by the Numbers
Explore NSF's investments in science and engineering across the nation and in your state by fiscal year.
NSF Funded Projects (Awards)
Explore our database of funded projects to learn what we're doing across the U.S.
NSF Fact Sheets
Learn about NSF's investments in your state and how the agency is catalyzing innovation in emerging technologies.
NSF Public Repository
Explore NSF-funded scholarly publications and datasets.