News Release 06-059
New Key Fits Old Lock
Researchers document step-by-step genetic changes affecting the structure and function of ancient proteins

Scientists recently demonstrated the evolution of two specific hormonal regulation systems.
April 7, 2006
This material is available primarily for archival purposes. Telephone numbers or other contact information may be out of date; please see current contact information at media contacts.
Looking back in time some 450 million years, researchers at the University of Oregon have reconstructed the evolution of two hormone molecules and their "receptors": the precisely shaped protein molecules that allow the body's cells to respond to the hormones.
Writing in the April 7 issue of the journal Science, a team led by Oregon biologist Joseph Thornton trace the stepwise genetic changes that allowed a single ancient ancestor molecule to produce two different modern receptors: one for the hormone aldosterone, which regulates kidney, colon and brain function, and the other for the hormone cortisol, which regulates the body's long-term response to stress.
To achieve this reconstruction, Thornton's group first characterized the receptors of several ancient vertebrate species, including lampreys, hagfish, and skates. Then, by applying standard statistical methods, the researchers showed that the two receptors found in most present-day vertebrates likely originated from a gene duplication that occurred about 450 million years ago. Finally, they inferred the gene sequence of the ancient receptor, synthesized it in the laboratory, and characterized its functions.
Their experiments showed that this synthesized receptor bound to an ancient hormone called deoxycorticosterone, as well as to the similarly shaped aldosterone and cortisol molecules. Thornton was surprised by the result, as aldosterone and cortisol did not exist until millions of years later. In effect, the new keys fit the old lock - opening the door to novel functions.
Thornton's group also identified two mutations--which presumably occurred millions of years after the duplication event--that enhanced the specificity of the modern-day receptor for cortisol.
Thornton said, "New techniques allowed us to see how ancient genes and their functions evolved hundreds of millions of years ago. We found that complexity evolved piecemeal with old genes being recruited to participate in new interactions and functions."
Thornton's research was supported in part by the National Science Foundation (NSF). William Zamer, the NSF program director who oversees the project commented, "Thornton's work is important scientifically because it shows how computational approaches can be used to infer the structure of ancient molecules, providing us with a window on how evolution has worked. Furthermore, it provided evidence that even highly specific receptor-hormone systems are not irreducibly complex and can come into existence through processes of Darwinian evolution."
In addition to NSF, the National Institutes of Health and the Alfred P. Sloan foundation supported the research.
For more on this story see the University of Oregon's news release.
-NSF-
-
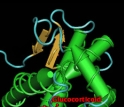
The molecular structure of two hormone receptors is shown.
Credit and Larger Version
Media Contacts
Richard (Randy) Vines, NSF, (703) 292-7963, email: rvines@nsf.gov
Program Contacts
William E. Zamer, NSF, (703) 292-8421, email: wzamer@nsf.gov
Principal Investigators
Joseph Thornton, University of Oregon, (541) 346-0328, email: joet@uoregon.edu
Related Websites
University of Oregon news release: http://www.uoregon.edu/newsstory.php?a=4.6.06-Thornton-irreducible-complexity.html
The U.S. National Science Foundation propels the nation forward by advancing fundamental research in all fields of science and engineering. NSF supports research and people by providing facilities, instruments and funding to support their ingenuity and sustain the U.S. as a global leader in research and innovation. With a fiscal year 2023 budget of $9.5 billion, NSF funds reach all 50 states through grants to nearly 2,000 colleges, universities and institutions. Each year, NSF receives more than 40,000 competitive proposals and makes about 11,000 new awards. Those awards include support for cooperative research with industry, Arctic and Antarctic research and operations, and U.S. participation in international scientific efforts.
Connect with us online
NSF website: nsf.gov
NSF News: nsf.gov/news
For News Media: nsf.gov/news/newsroom
Statistics: nsf.gov/statistics/
Awards database: nsf.gov/awardsearch/
Follow us on social
Twitter: twitter.com/NSF
Facebook: facebook.com/US.NSF
Instagram: instagram.com/nsfgov