All Images
Research News
Nanotubes Not for Toothpaste . . . Yet
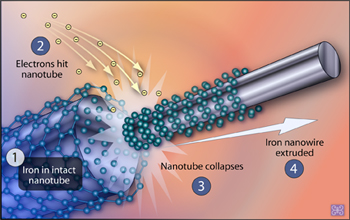
When a beam of electrons hits a carbon nanotube, some carbon atoms are knocked away, causing the remaining atoms to contract and the tube to shrink. When the tube is filled with even incredibly hard substances like iron carbide, the contraction squeezes the substance out as ultra-thin wires.
Credit: Zina Deretsky, National Science Foundation
Download the high-resolution JPG version of the image. (368 KB)
Use your mouse to right-click (Mac users may need to Ctrl-click) the link above and choose the option that will save the file or target to your computer.

Electron micrographs reveal the patterned atomic structure of the carbon nanotube (light grey) and the iron-carbide interior material (dark grey). Image 1 depicts the nanotube before contraction; image 2 shows material compressing during contraction. Image 3 shows how the wire pinches off at the conclusion of the process.
Credit: F. Banhart, University of Mainz, Germany
Download the high-resolution JPG version of the image. (98 KB)
Use your mouse to right-click (Mac users may need to Ctrl-click) the link above and choose the option that will save the file or target to your computer.


